At age 49, Jan Janisch-Hanzlik’s multiple sclerosis was destroying her freedom to live the life she wanted. She gave up her active nursing job for a desk role. Frequent falls made her afraid to carry her grandchildren. She had to move to a bigger house to make room for the wheelchair she feared she might end up needing full-time.
Even the best available medication wasn’t improving Janisch-Hanzlik’s symptoms, and she worried they’d only get worse. So when she learned about a trial of CAR T cell therapy at the University of Nebraska Medical Center in Omaha, close to the city of Blair where she lives, she phoned the clinic every other month until they were ready to enroll her as the first patient.
Originally designed to target and wipe out cancer by reprogramming the patient’s immune cells, CAR T is now being offered to patients in hundreds of clinical trials for autoimmune conditions like multiple sclerosis, lupus, Graves’ disease, vasculitis, and many others. The hope is that CAR T can duplicate the success it has demonstrated in a range of blood cancers by hunting down and eliminating cells that target the self in autoimmune diseases. This would essentially reset the body’s defenses to a state like the one that existed before the disease took hold.
But along with CAR T’s promise come risks, questions, and challenges. There’s uncertainty about how well it will work for autoimmunity and how long any benefits might last, as well as what long-term side effects might arise. Janisch-Hanzlik knew this when she sat down to receive the experimental treatment on June 9, 2025; she felt a mix of hope and fear knowing that she would be spending the next week being monitored for side effects including dangerous inflammation.
In addition to her clinical expertise and desire to pioneer a new treatment, Janisch-Hanzlik’s two young grandchildren helped inspire her pursuit of a treatment with known risks and uncertain benefits. Because multiple sclerosis has a genetic component, Janisch-Hanzlik knew that they have an elevated chance of going through the same struggle she has. “I would want to be able to say I did everything that I possibly could to prevent them, or anyone else, from having something like this,” she says.
From cancer to autoimmunity
The first CAR T cancer treatment was approved by the Food and Drug Administration in 2017 for an aggressive form of leukemia. Since then, the powerful and intensive treatment has resulted in long-term remission for many cancer patients.
The basic premise of CAR T is to activate the power of key immune cells called T cells. T cells normally recognize other cells that have been infected by a virus or bacterium, or are otherwise abnormal, and either destroy them or recruit other parts of the immune system to do so.
In CAR T for cancer, scientists engineer those T cells to specifically hunt and destroy malignant cells. The technology got its start when cancer researchers figured out how to take out a patient’s own T cells, insert DNA instructions for a “chimeric antigen receptor,” or CAR, and put them back into the person’s circulation. The CAR, which sits on the T cell’s surface and latches on to a specific molecular partner on the surface of cancerous cells, activates the T cell to attack.
Today CAR T cells are most commonly programmed to attack B cells, another key immune player. B cells are normally responsible for making antibodies, but in certain blood cancers, they proliferate out of control. By giving T cells a CAR that recognizes one of a couple of molecules unique to the B cell surface, the cells are reprogrammed to find and eliminate those cancerous cells.
B cells also are the central problem in many autoimmune conditions: They mistakenly make antibodies against normal tissues instead of against invading pathogens. So as CAR T began to succeed against B cell cancers, it didn’t take long for doctors to reason that CAR T therapy might also be able to wipe out bad B cells in people with autoimmunity.
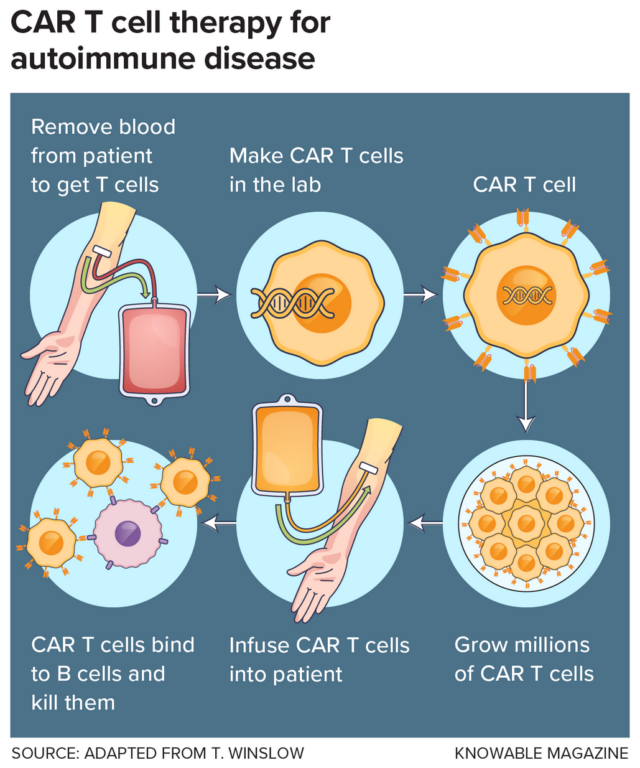
A German team pioneered autoimmune CAR T in a woman with lupus, reporting positive results in 2021. Since then, that team and others have worked to translate the oncology success of CAR T to tackle a broad spectrum of autoimmune diseases.
“I think it’s a game changer,” says Amanda Piquet, an autoimmune neurologist at the University of Colorado Anschutz in Aurora. Piquet is evaluating CAR T therapy for a rare and poorly understood autoimmune condition called stiff person syndrome, with symptoms including muscle stiffness and painful spasms. There is no FDA-approved treatment. When she heard about a company called Kyverna that was testing CAR T cell therapy in the syndrome, she thought it was “a perfect opportunity.”
The study she led, which reported preliminary results in December 2025, tested a single dose of CAR T in 26 people. Before the treatment, many participants struggled with a slow, mechanical gait, and 12 used assistive devices such as walkers and canes. Most patients were able to walk faster by 16 weeks post-treatment, and eight no longer needed their assistive devices for short distances. In April, the company reported that all 26 patients, as of their last follow-up appointment four to 12 months out from the therapy, were no longer using any other immunotherapies.
Risks and uncertainties
Despite such striking results, reprogramming the immune system is no simple matter. In early treatment of cancer patients, CAR T cells produced life-threatening side effects, as outlined in a 2026 article in the Annual Review of Medicine. As CAR T cells attack their targets, the associated inflammation can cause symptoms like high fevers and low blood pressure. If that inflammation reaches the brain, it can cause additional problems such as confusion and drowsiness.
Fortunately, physicians now have a decade’s worth of experience recognizing and treating these problems. “They’re certainly reversible and don’t cause long-term damage most of the time,” says Emily Littlejohn, a rheumatologist at the Cleveland Clinic.
Physicians and patients also must contend with decreased immunity as both a side effect of the treatment and its desired outcome. In CAR T treatment, doctors typically use powerful chemotherapy drugs to temporarily reduce the body’s immune cell population to make room for the new, engineered cells, leaving patients temporarily immunosuppressed. And if the treatment works, it will decimate B cell populations. Patients can be vulnerable to infections for up to a year after treatment, says Littlejohn.
These effects are manageable with preventive antibiotics, antivirals and vaccines. Patients also retain antibodies that their B cells made before the treatment, which provide residual protection for a few months. And for reasons that are not yet fully understood, CAR T seems to leave older B cells, which provide immune memory of past infections, intact in some cases. One study found that autoimmune patients treated with CAR T still made antibodies for diseases they’d been previously vaccinated against, like chicken pox and measles. These are signs that the treatment did not completely return the immune system to its factory settings.
When evaluating CAR T risk, it’s important to consider that many existing treatments for autoimmune disease also suppress the immune system for as long as a person takes them, experts note.
But there are other possible CAR T risks for autoimmune patients. In February, FDA officials published a paper endorsing CAR T’s potential in autoimmunity but warning of “unpredictable long-term toxicity.” CAR T treatment for cancer, the authors noted, has been linked to diverse long-term issues such as Parkinson’s disease. There have also been cases where the bioengineered cells themselves turned malignant, causing new, T cell-based cancers.
Causing a secondary cancer may be an acceptable risk when treating a life-threatening cancer, but probably not for autoimmunity, says Matt Lunning, medical director for gene and cellular therapy at Nebraska Medicine in Omaha. How to balance the risk between the impacts of an autoimmune disease, which can range widely in severity, and the difficult-to-quantify risk of future side effects or cancers remains a major open question.
Researchers are already working on second- and third-generation versions of CAR T that they expect to be safer for both cancer and autoimmunity. For example, James Howard, a neuromuscular neurologist at the University of North Carolina at Chapel Hill, is testing a technology from a company called Cartesian Therapeutics that encodes the CAR using molecules of mRNA, the short-lived genetic messenger used in Covid-19 vaccines, instead of long-lasting DNA. The CAR T cells should wipe out B cells for only as long as the mRNA persists, then lose their B cell-targeting abilities. With no chance for genetically modified T cells to hang around long-term, there should be no cancer risk.
Another plus of Cartesian’s approach: Physicians infuse these T cells in sufficient numbers that they don’t need to reproduce in the patient’s body, which Howard thinks reduces risk for inflammation. In a recent trial, 15 people with autoimmune diseases received the Cartesian CAR T treatment; two-thirds saw their symptoms improve, and none suffered long-term serious side effects.
Treating CAR T sticker shock
Beyond side effects, the other major challenge facing CAR T therapy is its price tag, which reaches hundreds of thousands of dollars including hospital stays, cell engineering, and other expenses.
The treatment would likely be cheaper, and simpler, if scientists could eliminate the need for personalized engineering of each patient’s own cells and instead use donor cells, or if they could cut out the step of engineering and growing the cells in a laboratory. Lunning says he is eyeing up-and-coming procedures that would modify a person’s T cells within their own body instead of doing the genetic engineering in a lab.
Researchers are even further along with a version of CAR T that uses healthy donors as a source of T cells. These could then be used by many patients in an “off-the-shelf” approach. It’s a method that has its own challenges, because of the immune mismatch between donor and patient cells that would lead them to attack each other. This problem can be overcome with additional genetic modifications to the donated T cells that prevent recipient and donor systems from recognizing each other as foreign, says Bing Du, an immunologist at East China Normal University in Shanghai who’s among many researchers working on this approach. Du estimates a lab could make CAR T cells for more than 1,000 patients from a single donor’s blood cells, at significant cost savings.
This kind of off-the-shelf CAR T therapy is what Janisch-Hanzlik of Nebraska received, under Lunning’s care, in 2025. The study organizers at TG Therapeutics expect to complete their research in early 2029.
Janisch-Hanzlik ended up sailing through the follow-up without side effects. A couple of months after the infusion, she was watching TV when she noticed she no longer needed special glasses to correct double vision. She started forgetting to bring her cane when moving about her house or going grocery shopping; she didn’t need it. Now, nearly a year since the treatment, she rarely falls and no longer requires a daily, three-hour nap. She recently enjoyed a trip to the Grand Canyon and looks forward to spending more time with her grandchildren.
She does still have symptoms, including weakness in her right leg, numbness and tingling in her feet, and difficulty finding the right word when speaking. She asks her doctors if they think she’s going to get better, stay the same, or get worse again.
“I have been told so many times, ‘We don’t know, you’re the first. We’re just going to have to wait and see,’” she says. “I definitely am thankful for every day I have.”
This article originally appeared in Knowable Magazine, a nonprofit publication dedicated to making scientific knowledge accessible to all. Sign up for Knowable Magazine’s newsletter.